Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Iida, Yoshihisa; Barr, L.; Yamaguchi, Tetsuji; Hemmi, Ko
Genshiryoku Bakkuendo Kenkyu (CD-ROM), 23(1), p.3 - 8, 2016/06
Thorium (Th)-229 is one of the important radionuclides for the performance assessment calculations for high-level radioactive waste repositories. The sorption behavior of Th onto montmorillonite and illite were investigated by batch sorption experiments. Experiments were carried out under variable pH and carbonate concentrations. The sorbability of montmorillonite was higher than that of illite. Distribution coefficients, 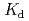 (m
(m kg
kg ), decreased with increased carbonate concentrations and showed the minimal value at around pH 10. The sorption behaviors of Th were analyzed by the non-electrostatic surface complex model with PHREEQC computer program. The model calculations were able to explain the experimental results reasonably well. The decreases of
), decreased with increased carbonate concentrations and showed the minimal value at around pH 10. The sorption behaviors of Th were analyzed by the non-electrostatic surface complex model with PHREEQC computer program. The model calculations were able to explain the experimental results reasonably well. The decreases of 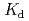 was likely due to the stabilization of aqueous species by hydroxo-carbonate complexations in the solutions.
was likely due to the stabilization of aqueous species by hydroxo-carbonate complexations in the solutions.
Futakuchi, Katsuhito*; Hashimoto, Shuji*; Sakuramoto, Yuji*; ;
JNC TN8400 2001-007, 52 Pages, 2001/04
As a natural analogue, the authors investigted a Tertiary argillaceous bed and a Quarternary hypabyssal rock (porphyrite) which intruded into the argillaceous rock, distributed in the Nishikubiki district of Niigata prefecture in Japan. We examined the variation of clay mineral species in the argillaceous rock surrounding the intrusive rock and carried out thermal analyses for the argillaceous rock based on the coolig history of the intrusive rock. The predominant clay mineral varied from montmorillonite to illite through illite/montmorillonite interlayers with approaching to the intrusive rock. The thermal analyses indicated that the temperature descended from 270 to 15  C during the 7.5
C during the 7.5 10
10 years at alocalty of argillaceous rock containing 75% illite in the interlayers. On the assumption that the alteration from montmorillonite to illite was regarded as a first-order reaction, we evaluated the apparent activation energy based on the thermal condition mentioned above; about 103 kJ/mol was obtained for this illitization. This was within the range of values reported previously by laboratory experiments and/or examinations of natural illitizations.
years at alocalty of argillaceous rock containing 75% illite in the interlayers. On the assumption that the alteration from montmorillonite to illite was regarded as a first-order reaction, we evaluated the apparent activation energy based on the thermal condition mentioned above; about 103 kJ/mol was obtained for this illitization. This was within the range of values reported previously by laboratory experiments and/or examinations of natural illitizations.
Imai, Jun*
JNC TJ8400 2000-008, 196 Pages, 2000/02
The objective of this research is to make clear long-term alteration processes of bentonite contacting with concrete under a repository condition for radioactive waste. The Uzu tunnel in yamagata prefecture in Japan, constructed during the term of December of 1963 to July 1967, was selected as an appropriate natural analogue: the tunnel wall was made of portland cement and which has been contacting with a bentonite bed during  32 years. Sample analyses indicated that the original bentonite was Na
32 years. Sample analyses indicated that the original bentonite was Na -type and it changed to Ca
-type and it changed to Ca -type in the range of a few millimeters from the contact. Although a Ca
-type in the range of a few millimeters from the contact. Although a Ca leaching was also observed from the concrete near the contact, neither transformation to zeolite nor to illite was recognized. On the other hand, sulfur increased and ettringite (3CaO
leaching was also observed from the concrete near the contact, neither transformation to zeolite nor to illite was recognized. On the other hand, sulfur increased and ettringite (3CaO  Al
Al O
O
 3CaSO
3CaSO 4
4  32H
32H O) was recognized in the concrete within the depth about 30 mm from the contact.
O) was recognized in the concrete within the depth about 30 mm from the contact.
Nagasaki, Shinya*
JNC TJ8400 2000-004, 32 Pages, 2000/02
Equilibrium and kinetics of sorption of NpO on illite were investigated at pH = 6 by using the differential pulse anodic stripping voltammetry method and the spectroscopic method, respectively. It was found that the sorption isotherm obtained was fitted better by the Langmuir-Freundlich type equation than by the Langmuir equation. The heterogeneity coefficient was 0.89
on illite were investigated at pH = 6 by using the differential pulse anodic stripping voltammetry method and the spectroscopic method, respectively. It was found that the sorption isotherm obtained was fitted better by the Langmuir-Freundlich type equation than by the Langmuir equation. The heterogeneity coefficient was 0.89  0.05 and the half width at half maximum (HWHM) of affinity spectrum was 0.19 log unit, indicating that the surfacc of illite used has a low degree of heterogeneity. The kinetic spectra indicated that the sorption of NpO
0.05 and the half width at half maximum (HWHM) of affinity spectrum was 0.19 log unit, indicating that the surfacc of illite used has a low degree of heterogeneity. The kinetic spectra indicated that the sorption of NpO occurs only at the outer surfacc. The mean HWHM of the kinetic spectra was 0.18 log unit. This also proves that the sorption kinetics of NpO
occurs only at the outer surfacc. The mean HWHM of the kinetic spectra was 0.18 log unit. This also proves that the sorption kinetics of NpO on the illite used is controlled by the same heterogeneity of the sorption sites. From the dependence of mean rate constants on temperature, a mean apparent activation enthalpy and a mean apparent activation entropy were evaluated at 37
on the illite used is controlled by the same heterogeneity of the sorption sites. From the dependence of mean rate constants on temperature, a mean apparent activation enthalpy and a mean apparent activation entropy were evaluated at 37 3 kJ/mol and - 69
3 kJ/mol and - 69  7 J/K
7 J/K mol, respectively. This value of enthalpy suggests that the sorption is not controlled by diffusion through the hydrodynamic film around the illite. Equilibrium and kinetics of sorption of NpO
mol, respectively. This value of enthalpy suggests that the sorption is not controlled by diffusion through the hydrodynamic film around the illite. Equilibrium and kinetics of sorption of NpO and Np(V) carbonate complexes (mainly NpO
and Np(V) carbonate complexes (mainly NpO CO
CO ) on Na-montmorillonite were also examined by using same technique.
) on Na-montmorillonite were also examined by using same technique.
 thin films
thin filmsTanaka, Toru*; Wakahara, Akihiro*; Yoshida, Akira*; Oshima, Takeshi; Ito, Hisayoshi; Okada, Sohei
Journal of Applied Physics, 87(7), p.3283 - 3286, 2000/01
Times Cited Count:11 Percentile:46.92(Physics, Applied)no abstracts in English
Sato, Tsutomu; Murakami, Takashi*; Watanabe, Takashi*
Clays and Clay Minerals, 44(4), p.460 - 469, 1996/00
Times Cited Count:37 Percentile:75.43(Chemistry, Physical)no abstracts in English
Sato, Tsutomu; Murakami, Takashi*; Isobe, Hiroshi; Onuki, Toshihiko
Materials Research Society Symposium Proceedings, Vol.353, 0, p.239 - 246, 1995/00
no abstracts in English
Onuki, Toshihiko; Murakami, Takashi; Sato, Tsutomu; Isobe, Hiroshi
Radiochimica Acta, 66-67, p.323 - 326, 1994/00
no abstracts in English
Sato, Tsutomu
SMECTITE, 3(1), p.39 - 41, 1993/05
no abstracts in English
Murakami, Takashi; Sato, Tsutomu; Watanabe, Takashi*
American Mineralogist, 78(3), p.465 - 468, 1993/00
no abstracts in English
Onuki, Toshihiko; Murakami, Takashi; Sato, Tsutomu; Isobe, Hiroshi
Nihon Genshiryoku Gakkai-Shi, 34(12), p.1139 - 1142, 1992/12
Times Cited Count:0 Percentile:0.01(Nuclear Science & Technology)no abstracts in English
Kamei, Gento; Yusa, Yasuhisa; Sasaki, Noriaki
PNC TN8410 91-253, 8 Pages, 1992/01
Time-temperature conditions and water chemistry on illitization at the Murakami deposit in central Japan were determined. The extent of the illitization and time-temperature condition estimated were as follows: the duration for conversion from 0 to 80% illite (volumetric ratio) was approximately 3.5 Ma in the temperature range from 340 to 100 C, Conversion from 0 to 40% requires approximately 3.0 Ma in the temperature range from 240 to 100
C, Conversion from 0 to 40% requires approximately 3.0 Ma in the temperature range from 240 to 100 C, During 2.0Ma in the temperature range from 160 to 100
C, During 2.0Ma in the temperature range from 160 to 100 C, however, illite was scaroely observed, Water chemistry is estimated from two approaches, namely laboratory experiment and numerical analysis. The former is an interactive experiment between seawater and the tuff of the deposit. The latter is a calculation based on the difference of bulk composition between illitized and non-illitized tuff. The extent of each ionic concentration is inferred to be as follows: K
C, however, illite was scaroely observed, Water chemistry is estimated from two approaches, namely laboratory experiment and numerical analysis. The former is an interactive experiment between seawater and the tuff of the deposit. The latter is a calculation based on the difference of bulk composition between illitized and non-illitized tuff. The extent of each ionic concentration is inferred to be as follows: K ; 560 to 6400, Mg
; 560 to 6400, Mg ; 800 to 1700, Ca
; 800 to 1700, Ca ; 360 to 2900, Na
; 360 to 2900, Na ; 9400 to 15000 (mg/l).
; 9400 to 15000 (mg/l).